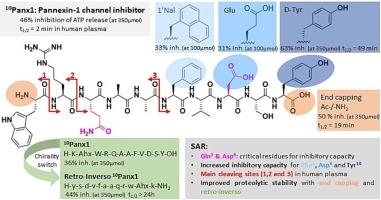
Pannexin1 channels facilitate paracrine communication and are involved in a broad spectrum of diseases. At- tempts to find appropriate pannexin1 channel inhibitors that showcase target-selective properties and in vivo applicability remain nonetheless scarce. However, a promising lead candidate, the ten amino acid long peptide mimetic 10Panx1 (H-Trp1-Arg2-Gln3-Ala4-Ala5-Phe6-Val7-Asp8-Ser9-Tyr10-OH), has shown potential as a pan- nexin1 channel inhibitor in both in vitro and in vivo studies. Nonetheless, structural optimization is critical for clinical use. One of the main hurdles to overcome along the optimization process consists of subduing the low biological stability (10Panx1 t1/2 = 2.27 ± 0.11 min). To tackle this issue, identification of important structural features within the decapeptide structure is warranted. For this reason, a structure–activity relationship study was performed to proteolytically stabilize the sequence. Through an Alanine scan, this study demonstrated that the side chains of Gln3 and Asp8 are crucial for 10Panx1′s channel inhibitory capacity. Guided by plasma stability experiments, scissile amide bonds were identified and stabilized, while extracellular adenosine triphosphate release experiments, indicative of pannexin1 channel functionality, allowed to enhance the in vitro inhibitory capacity of 10Panx1.
