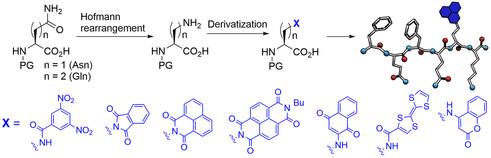
We present a convenient and efficient approach for synthesizing lysine homologs via the Hofmann rearrangement of N-protected
L-asparagine and L-glutamine. This method enabled the preparation of a diverse family of noncanonical amino acids (ncAAs)
bearing side chains with varied properties, including electron-donating and electron-accepting abilities, redox activity, and fluorescence.
The luminescence properties and redox behavior of these ncAAs were investigated. Furthermore, the newly synthesized
Fmoc-protected ncAAs were successfully incorporated into a hexapeptide sequence using solid-phase peptide synthesis, thus
expanding the library of hexapeptide hydrogelators.
Link to Publisher’s page: Hofmann Degradation of Asparagine and Glutamine as an Efficient Approach for the Synthesis of Derivatized Lysine Homologs - ScienceDirect
