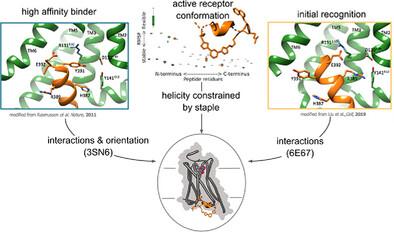
G protein-coupled receptors (GPCRs) are involved in most human physiological processes and one of the largest families of approved drug-targeted proteins. Heterotrimeric Gαβɣ proteins bind to the intracellular cavity of the activated receptor mainly through the C-terminal α5 helix of the Gα subunit (GαCT). Modulation of GPCR activity through intracellular GPCR binding sites is emerging. Here we develop highly active GαsCT-derived peptidomimetics that stabilize the β2 adrenergic receptor (β2AR) in an active-like conformation when the helical conformation of GαsCT is preserved by a covalent tether ("staple"). By rational design, integrating the information of two crystal structures showing different binding modes of GαsCT with β2AR, an appropriate staple position was identified. The key interactions observed in the two distinct β2AR structures (β2AR-Gs(empty)) and β2AR-T4L-GsCT-CC), namely Y391 and E392, were substituted with non-canonical amino acids to improve activity. Optimization resulted in the identification of a potent peptidomimetic capable of stabilizing an active-like receptor conformation, whilst blocking receptor-mediated cAMP formation. Molecular dynamics simulations indicated a peptidomimetic binding mode that may represent another intermediate state preceding that of β2AR-Gs(empty). We envision this approach to be useful for further structural and functional exploration of other GPCRs or as a tool in drug discovery.
Nano‐Gs Protein Peptidomimetics: Rational Design of Gα C‐Terminus‐Derived Peptides Mimicking Key Components of Gs‐β2AR Interactions - Tran - Angewandte Chemie International Edition - Wiley Online Library
