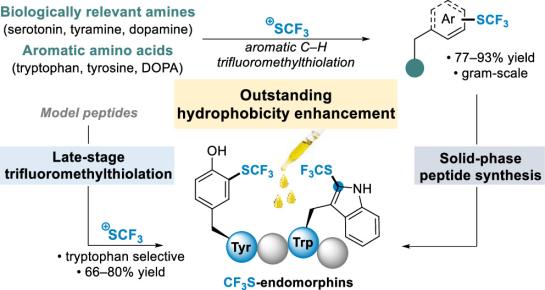
The incorporation of fluorinated amino acid residues into peptides represents a promising strategy for improving the
pharmacokinetic properties of bioactive peptides. Herein, we report the solid-phase synthesis (SPPS) of a set of ten SCF3- or CF3-
modified neuropeptide analogs based on endomorphin-1 (EM1). EM1, a selective subnanomolar μ-opioid receptor (μOR) agonist
with poor metabolic stability (t1/2 = 6 min in human plasma), was selected as a model to evaluate the effects of SCF3 and CF3
functionalization on the pharmacokinetic profile of short bioactive peptides. The syntheses of eight ready-to-use SCF3- or CF3-
containing building blocks for SPPS and their incorporation into peptides are reported. In vitro μOR binding and functional activity
assays demonstrated that most fluorinated analogs retained binding affinity and potency, accompanied by increased hydrophobicity.
Among the series, modification of the pharmacophoric Tyr1 residue with L-Dmt(3-SCF3) yielded the most favorable profile (Ki = 1.4
nM, EC50 = 0.9 nM). Plasma stability studies revealed a significant increase in half-life for this ligand (72-fold relative to EM1 and
14-fold relative to EM1(Dmt1)), and thus, further demonstrated the potential of SCF3-containing amino acids in therapeutic peptide
design.
Link to Publisher’s page: Trifluoromethylthio and Trifluoromethyl Functionalization of Endomorphin-1 Enhances its Hydrophobicity and Plasma Stability while Preserving its Affinity for the μ-Opioid Receptor | The Journal of Organic Chemistry
